- Home
- compression factor equation
- What is the value of compression factor Z for the gas? (A) 1 (B) >1 (C) <1 (D) Zero
What is the value of compression factor Z for the gas? (A) 1 (B) >1 (C) <1 (D) Zero
5 (83) · $ 14.50 · In stock

Walpole 8 probabilidad y estadística para ciencias e ingenierias

The compressibility factor Z a low-pressure range of all gases except hydrogen is:Z=(1+ displaystylefrac{a}{V_{m}RT})Z=(1 -displaystylefrac{a}{V_{m}RT})Z=(1+displaystylefrac{Pb}{RT})Z = ( 1 - displaystylefrac{Pb}{RT})
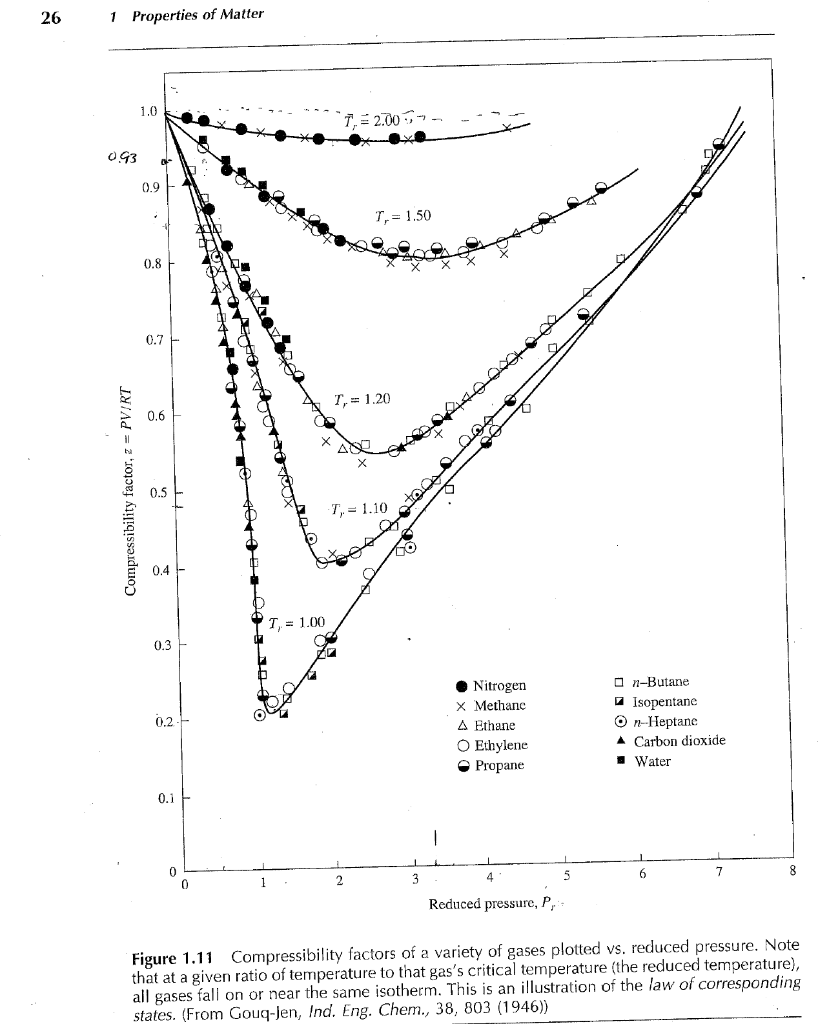
Solved 3) Using the “Z graph” to estimate the compression

AISC SEISMIC PROVISIONS FOR STRUCTURAL STEEL BUILDINGS by ALFREDO

Thermodynamics An Engineering Approach 6th Edition Cengel Solutions Manual by Desiree - Issuu

Solved The virial expansion of the compression factor (Z)

3.2 Real gas and compressibility factor – Introduction to

Compressibility factor, Z of a gas is given as Z= frac { pV }{ nRT } (i) What is the value of Z an ideal gas?(ii) For real gas what will be

Real gas z-Factor chart [2] Download Scientific Diagram

P k nag solution by Shaikh Mohd Aslam - Issuu

Compressibility Factor Z
:format(webp)/https://static-ph.zacdn.com/p/dkny-6761-0979672-1.jpg)











